Viruses, Systemic Disease, and Dentistry
Understanding the problem and how to protect your practice
William D. Nordquist DMD MS, William C Domb DMD
Viruses and periodontal disease
Recent study is now pointing toward viruses as an extremely important component of periodontal disease. Both in initiation as well as ongoing promotion. Several years ago, Jorgen Slots, DDS, PhD, Professor of Dentistry from the University of Southern California lectured that herpesviruses [both Epstein-Barr virus type 1 (EBV-1) and human cytomegalovirus (HCMV)] are some unrecognized major viruses implicated1. Unfortunately, when one transitions from the study of bacteria to viruses, most dentists cringe, since we have the least amount of training in comprehending that submicroscopic world.
This short monograph will present a compact view of viruses and their oral impacts. And viruses, perhaps initially stemming from the mouth may have much wider systemic impacts. Ruth F. Itzhaki2 reported conclusive evidence that demonstrates the Herpes simplex virus type 1 (HSV1), the very same virus that causes cold sores in the mouth, are present in the elderly human brain. HSV1 infects the epithelial cells of the mucous membranes of the face where it secondarily infects sensory nerve terminals. Once within the neuron, HSV1 travels back to the neuronal cell body located in the trigeminal ganglion, which lies at the base of the skull adjacent to the brain stem. HSV1 remains in the ganglion in a latent form, until reactivation when newly synthesized virus is transported back more peripherally3. As proposed by Itzhaki, this highly prevalent virus reactivates and enters the brain in older age by way of these peripheral nervous
system or the olfactory route4. HSV1becomes latent in the brain, but periodically reactivates in association with systemic infection, immunosuppression, or other stressors.
Spirochetes and activation of HSV1
Pathogen-induced infla mmation and central nervous system accumulation of Beta Amyloid(Aβ) (Figure 1) damages the blood-brain barrier, which contributes to the pathophysiology of Alzheimer’s Disease (AD)5. Data for both C. pneumoniae and spirochetes6 7 shows a high prevalence of these pathogens in AD brains only, suggesting that secondary C. pneumoniae and/or spirochete infection of brain may occur after preliminary HSV1, and other co-factors have already initiated AD pathogenesis8. Since spirochetes can invade all tissue of the body via blood vessel pathways9, Nordquist suggests spirochetes may be the instigators of the inflammation that damages the lining of blood vessels and possibly deteriorates the blood brain barrier of the brain10. Spirochetes can also take the Trigeminal nerve pathway to the brain11. Thus, the activation of the already present latent HSV1 by spirochetes in the brain could represent the final step in a series of infections leading to full blown Alzheimer’s disease.
mmation and central nervous system accumulation of Beta Amyloid(Aβ) (Figure 1) damages the blood-brain barrier, which contributes to the pathophysiology of Alzheimer’s Disease (AD)5. Data for both C. pneumoniae and spirochetes6 7 shows a high prevalence of these pathogens in AD brains only, suggesting that secondary C. pneumoniae and/or spirochete infection of brain may occur after preliminary HSV1, and other co-factors have already initiated AD pathogenesis8. Since spirochetes can invade all tissue of the body via blood vessel pathways9, Nordquist suggests spirochetes may be the instigators of the inflammation that damages the lining of blood vessels and possibly deteriorates the blood brain barrier of the brain10. Spirochetes can also take the Trigeminal nerve pathway to the brain11. Thus, the activation of the already present latent HSV1 by spirochetes in the brain could represent the final step in a series of infections leading to full blown Alzheimer’s disease.
Figure 1. Amyloid beta
Antiviral properties of β-amyloid
Amyloid plaques, the hallmark of AD, contain fibrillar Aβ. HSV1 has been implicated as a risk factor for AD and found to co-localize within Aβ  plaques. Aβ peptides represent antimicrobial peptides that protect against neurotropic virus infections such as HSV1 (Figure 3). The Aβ peptide may protect against latent herpes viruses and against other infections. This antimicrobial property may explain why Aβ plaque formation plays a pathogenic role in the progression of the sporadic form of AD because it is reacting to an infection12 13.
plaques. Aβ peptides represent antimicrobial peptides that protect against neurotropic virus infections such as HSV1 (Figure 3). The Aβ peptide may protect against latent herpes viruses and against other infections. This antimicrobial property may explain why Aβ plaque formation plays a pathogenic role in the progression of the sporadic form of AD because it is reacting to an infection12 13.
Aβ plaque in Alzheimer’s disease is association with active biofilms of Borrelia Burgdorferi (the Lyme disease spirochete)
Very recent research has shown that Aβ plaque in Alzheimer’s disease is an active biofilm of active Borrelia spirochetes in Alzheimer’s disease patients. MacDonald presented compelling evidence from brain tissue harvested from deceased Alzheimer’s patients. In this 37-minute video14, Borrelia
Figure 2 fibrillar β-amyloid (Aβ).
Figure 3 shows the Herpes Simplex Virus showing icosahedral capsid that resembles the bacteriophage capsid.
Chronic Brain Infections and Development of Alzheimer’s Disease, MacDonald used specific DNA marker probes to identify Borrelia Burgdorferi (implicated in Lyme diseas e) spirochetes, if present. 100% of samples tested demonstrated that active biofilms of various forms of Borrelia were enveloped within Aβ. MacDonald, for the very first time, demonstrated the spore-like role of granular forms of Borrelia, as viable, virulent pathogens, distinct from the spiral Borrelia corkscrew shaped forms. He identified round bodies forms of spirochetes in the evolution of Alzheimer’s disease. All these forms live within active biofilm communities previously characterized as undefined dead brown appearing lesions called Aβ plaques.
e) spirochetes, if present. 100% of samples tested demonstrated that active biofilms of various forms of Borrelia were enveloped within Aβ. MacDonald, for the very first time, demonstrated the spore-like role of granular forms of Borrelia, as viable, virulent pathogens, distinct from the spiral Borrelia corkscrew shaped forms. He identified round bodies forms of spirochetes in the evolution of Alzheimer’s disease. All these forms live within active biofilm communities previously characterized as undefined dead brown appearing lesions called Aβ plaques.
The roles for Aβ in diabetes and Alzheimer’s disease
Growing evidence is showing that diabetes and Alzheimer’s disease are concurrent diseases. Plaques containing fibrillar Aβ deposits associated with dying cells and inflammatory processes are hallmark pathological features in Alzheimer’s disease as well as diabetes15 16. The accumulation of Aβ within islet β-cells is a major pathological feature of the pancreas in patients with chronic diabetes.
The role of viruses in Creutzfeldt–Jakob disease the human form of Mad Cow disease
Viruses may be involved Creutzfeldt–Jakob disease (CJD)17despite its earlier connection with prion accumulation. CJD is usually referred to by the more commonly known name of the human form of “mad cow disease” [bovine spongiform encephalopathy (BSE)]18. This devastating disease is a degenerative neurological disorder that is irrepressible and inevitably fatal. However, given that BSE is believed to be the same variant as CJD19 in humans, the two are often almost identical.
CJD is characterized by the presence of a deformed protein called a prion. There is controversy as to how the disease progresses. The explanation most commonly accepted is that a defective prion replicates by converting its properly folded counterparts, in the host, to the same misfolded structure it possesses, “prion only theory”20. Another theory proposed by Laura Manuelidis21 proposed that virus particles cause CJD.22 Manuelidis claimed that although much work remains to be done, there is a reasonable explanation that viral particles cause the transformation of the prion that is the hallmark of CJD. It is likely that all the various versions of abnormal prion are the result of infection by an exogenous and stable viral particle, and are a consequence of the neurodegenerative disease process rather than its cause23. CJD causes the brain tissue to degenerate rapidly and, as the disease destroys the brain, the brain develops holes that resembles a sponge in appearance. Viruses can alter normal prions to fold into mutant prions.
To understand the significance of possible link between CJD and a virus in our discussion of viruses, systemic disease, and dentistry is the possibly that other neurologic diseases causing similar brain damage may also by caused by a virus. Alzheimer’s disease, Multiple sclerosis, Autism, Parkinson’s disease, Lou Gehrig’s disease Amyotrophic lateral sclerosis (ALS), and other neurological diseases have similar brain damage.
Alzheimer’s disease and viruses
Alzheimer’s disease24 is the most common of many neurodegenerative disorders primarily affecting ageing humans. Dohler, et al.25 reported an investigation that showed amyloid’s abnormal ability to attract and bind to prions, the same type of molecules that goes erratic in BSE and CJD. The main problem in Alzheimer’s Disease is that Aβ formation becomes so prevalent that this material interferes with normal electrical connections in the brain11. Normal amyloid is a stacking of normal prions26. Figure 4 show the difference between normal prion and the variant folded-over pathogenic form.
Aβ are peptides of 36–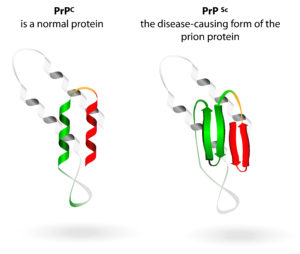 43 amino acids that are fatefully involved in Alzheimer’s disease as the main component of the Aβ plaques11. The peptides result from an amyloid precursor protein (APP), which is being altered by certain enzymes to yield Aβ. These molecules can cumulative to form flexible soluble Aβ fibrils. Abnormal Aβ is a variant binding of deformed amyloid with folded-over prions, the same problematic process that exists and is malformed in BSE and CJD.
43 amino acids that are fatefully involved in Alzheimer’s disease as the main component of the Aβ plaques11. The peptides result from an amyloid precursor protein (APP), which is being altered by certain enzymes to yield Aβ. These molecules can cumulative to form flexible soluble Aβ fibrils. Abnormal Aβ is a variant binding of deformed amyloid with folded-over prions, the same problematic process that exists and is malformed in BSE and CJD.
It is now believed that certain misfolded abnormal oligomers referred to as “seeds” can induce other amyloid molecules to also take a misfolded oligomeric form, leading to a chain reaction akin to an abnormal prion infection.27 The seeds, or the resulting Aβ plaques, are toxic to nerve cells. The other protein implicated in Alzheimer’s disease, tau protein, also forms prion-like misfolded oligomers, and there is some evidence that misfolded Aβ can induce tau to misfold.
In BSE and CJD, as reported earlier, there is a growing consensus that a virus is encoding via its aberrant preprogrammed DNA, to produce prions that are misfolded. If this hypothesis proves definitively to be true, then what is the possibility that the final step of the disease process in Alzheimer’s disease is also caused by the same virus?
Spirochetes and viruses
Spirochetes are associated with bacteria viruses called bacteriophages, normally lethal to bacteria. Eukaryotic dsDNA viruses (those viruses that replicate only inside the living cells) evolved from bacteriophages. The herpesviruses discussed earlier also evolved from a bacteriophage, but from a different evolutionary root. Papillomaviruses evolved from single-stranded DNA viruses and ultimately from plasmids28. When phages attach to and inject their DNA contents into spirochetes, they react differently from other bacteria and are not lysed by them29.
Instead, researchers realized that they could use these bacteria viruses as research tools30 to alter spirochetal DNA. Bacteriophages were used to inject virus DNA into spirochetes to increase their virulence, such as antibiotic resistance. The Borrelia burgdorferi spirochete is an alleged example of this potentially weaponizing alteration31. Bacteriophages have also been isolated in dental plaque32. Spirochetes contain plastids, which are small linear or circular extra-chromosomal, DNA. These packages can not only be transferred between other species spirochetes, but to other bacteria, as well in a process called transduction. Since bacteriophages and plastids are intimately associated with spirochetes (figure 6), it would not be much of a stretch to speculate that the herpesviruses are also associated with spirochetes since all three are so closely related to each other in their evolutionary tree. Spirochetes can transport these viruses by disseminating them to distant organs, further substantiating its potential importance in oral and linked systemic disease.
realized that they could use these bacteria viruses as research tools30 to alter spirochetal DNA. Bacteriophages were used to inject virus DNA into spirochetes to increase their virulence, such as antibiotic resistance. The Borrelia burgdorferi spirochete is an alleged example of this potentially weaponizing alteration31. Bacteriophages have also been isolated in dental plaque32. Spirochetes contain plastids, which are small linear or circular extra-chromosomal, DNA. These packages can not only be transferred between other species spirochetes, but to other bacteria, as well in a process called transduction. Since bacteriophages and plastids are intimately associated with spirochetes (figure 6), it would not be much of a stretch to speculate that the herpesviruses are also associated with spirochetes since all three are so closely related to each other in their evolutionary tree. Spirochetes can transport these viruses by disseminating them to distant organs, further substantiating its potential importance in oral and linked systemic disease.
Thus, it is possible that spirochetes serve as a rich source of transferable genetic material to remote location of the body. Since many spirochetal diseases produce neurological symptoms33, i.e., syphilis and Lyme disease. Further research is needed to evaluate whether herpesviruses or virus particles carried by spirochetes to the brain are associated with alteration of prions and Aβ.
 Figure 5 illustrates a bacteriophage. Notice the icosahedral capsid that resembles the herpes virus capsid.
Figure 5 illustrates a bacteriophage. Notice the icosahedral capsid that resembles the herpes virus capsid.
Figure 6 Bacteriophage injecting virus particles into a spirochete.
Prion Disease: The Implications for Dentistry
Reduce pathogen load
Since data supports the notion that spirochete infection may be the inflammatory stimulus that activates already present HSV-1 in the brain, logically eliminating this pathogen from the oral cavity would prevent its destructive cycle in AD. It is incumbent on dentists to diagnosis, reduce and/or eliminate the oral spirochetal pathogenic load from the diseased gingival sulcus. This can be done by many known periodontal treatment regiments followed by routine oral hygiene methods.
Reduce possibility of transmission between dental patients
Several articles have been written warning of possible transmission of prions from dental patients with CJD to other patients34 35 36. These articles emphasize the difficulty in sterilizing instruments contaminated by prions. Since dental pulp originates from richly innervated neural crest cells, it is theoretically possible that the dental pulp of individuals infected with CJD may be infectious37. Non-defective prion amyloid plaques in human periapical infected oral tissues have been associated with rheumatoid arthritis and chronic oral infections indicating antiviral activity38. These researchers suggest single use endodontic files as a precaution since cleaning and sterilization is very difficult, if not impossible.
Prion agents, unlike infectious microorganisms, resist conventional sterilization methods, such as steam autoclaving, even at increased temperatures, or by ethylene oxide gas39 40 41. Ozone gas can disrupt prions in seconds, so sterilizers using ozone as an adjunct have been put in use in Europe as well as some hospitals in the US.
Since the CJD is so rare, no case has been found that proves it can be transmitted from the use of contaminated dental instruments. However, warnings exist that a theoretical possibility exists for transmission to the extent that the United Kingdom has required strict guidelines in sterilization of instruments, health history, and management of these CJD patients. For these reasons, in the UK it is no longer permissible to reuse endodontic instruments with different patients. Prions have been detected in one in 250 people there.
The most recent literature has produced strong evidence that Alzheimer’s disease and diabetes, like CJD and BSE, also involves folded over prions and Aβ plaque. Since this revelation is so recent, to date, no research has been performed linking the spread of Alzheimer’s disease and diabetes prions between patients during dental procedures. However, the probability necessarily has increased exponentially since these two diseases represent a large percentage of the patient population. Further, new research has implemented herpesviruses and/or virus particles to prion formation. This fact also dramatically increases the probability that these viruses could be transferred between dental patients via contaminated instruments.
As suggested by Hill’s criteria, once the probability of a causal relationship between oral viruses, systemic disease and the brain, is established prompt action is needed. Stronger requirements for management of CJD, Alzheimer’s disease, and diabetic patients must be established to minimize transmission of these diseases among dental patients.
References:
1 Contreras A and Slots J. Herpesviruses in human periodontal disease. J Periodontal Res. 2000 Feb;35(1):3-16.
2 Itzhaki RF. Herpes simplex virus type 1 and Alzheimer’s disease: increasing evidence for a major role of the virus. Front Aging Neurosci. 2014 Aug 11;6:202.
3Bearer EL. Perspectives on herpes-APP interactions. Aging Cell. 2004 Apr;3(2):81-4.
4 Itzhaki RF, Cosby SL, Wozniak MA (2008) Herpes simplex virus type 1 and Alzheimer’s disease: The autophagy connection. J Neurovirol 14, 1-4.
5Harris SA, Harris EA. Herpes Simplex Virus Type 1 and Other Pathogens are Key Causative Factors in Sporadic Alzheimer’s Disease. J Alzheimers Dis. 2015;48(2):319-53.
6 Miklossy J (2011) Emerging roles of pathogens in Alzheimer disease. Expert Rev Mol Med 13, e30.
7 Riviere GR1, Riviere KH, Smith KS. Molecular and immunological evidence of oral Treponema in the human brain and their association with Alzheimer’s disease. Oral Microbiol Immunol. 2002 Apr;17(2):113-8.
8 Strandberg T, Pitkala K, Linnavuori K, Tilvis R (2003) Impact of viral and bacterial burden on cognitive impairment in elderly persons with cardiovascular disease. Stroke 34, 2126-2131.
9 Foschi F, Izard J, Sasaki H, Sambri V, Prati C, Müller R, Stashenko P. Treponema denticola in disseminating endodontic infections. J Dent Res. 2006 Aug;85(8):761-5.
10 Nordquist WD and Krutchkoff DJ. The Silent Saboteurs, The BioMed Publishing Group, Lake Tahoe, Nevada, 2010.
11 Riviere GR, Riviere KH, Smith KS. Molecular and immunological evidence of oral Treponema in the human brain and their association with Alzheimer’s disease. Oral Microbiol Immunol. 2002 Apr;17(2):113-8.
12 Bourgade K, Garneau H, Giroux G, Le Page AY, Bocti C, Dupuis G, Frost EH, Fülöp T Jr. β-Amyloid peptides display protective activity against the human Alzheimer’s disease-associated herpes simplex virus-1. Biogerontology. 2015 Feb;16(1):85-98.
13 Bourgade K, Dupuis G, Frost EH, Fülöp T. Anti-Viral Properties of Amyloid-β Peptides. J Alzheimers Dis. 2016 Oct 4;54(3):859-878.
14 MacDonald A. Review of Infectious Borrelia species Chronic Brain Infections and the Development of Alzheimer’s Disease. 2015 https://vimeo.com/145745120.
15 Riek R. Infectious Alzheimer’s disease? NATURE Vol 444|23 November 2006, 429-430.
16 Bharadwaj P, Wijesekara N, Liyanapathirana M, Newsholme P, Ittner L, Fraser P, Verdile G. The Link between Type 2 Diabetes and Neurodegeneration: Roles for Amyloid-β, Amylin, and Tau Proteins. J Alzheimers Dis. 2017;59(2):421-432.
17 https://en.wikipedia.org/wiki/Creutzfeldt%E2%80%93Jakob_disease.
18 https://en.wikipedia.org/wiki/Bovine_spongiform_encephalopathy.
19 https://en.wikipedia.org/wiki/Creutzfeldt%E2%80%93Jakob_disease.
20 Manuelidis L, Liu Y, Mullins B. Strain-specific viral properties of variant Creutzfeldt-Jakob disease (vCJD) are encoded by the agent and not by host prion protein. J Cell Biochem. 2009 Feb 1;106(2):220-31.
21 Manuelidis L. A 25 nm virion is the likely cause of transmissible spongiform encephalopathies J Cell Biochem. 2007 Mar 1;100(4):897-915.
22 Manuelidis L. Infectious particles, stress, and induced prion amyloids: a unifying perspective. Virulence. 2013 Jul 1;4(5):373-83. doi: 10.4161/viru.24838. Epub 2013 Apr 30.
23 Manuelidis L1, Liu Y, Mullins B. Strain-specific viral properties of variant Creutzfeldt-Jakob disease (vCJD) are encoded by the agent and not by host prion protein. J Cell Biochem. 2009 Feb 1;106(2):220-31.
24 https://www.nia.nih.gov/alzheimers/publication/alzheimers-disease-fact-sheet.
25 Dohler F, Sepulveda-Falla D, Krasemann S, Altmeppen H, Schluter H, Hildebrand D, Zerr I, Matschke L and Glatzel M. High molecular mass assemblies of amyloid-b oligomers bind prion protein in patients with Alzheimer’s disease. Brain. 2014 Mar;137(Pt 3):873-86.
26 Chen S, Yadav SP, and Surewicz WK. Interaction between Human Prion Protein and Amyloid-β (Aβ) Oligomers. J Biol Chem. 2010 Aug 20; 285(34): 26377–26383.
27 Gerson JE and Kayed R. Formation and Propagation of Tau Oligomeric Seeds. Front Neurol. 2013; 4: 93. Published online 2013 Jul 17. Prepublished online 2013 Jun 19.
28 Koonin EV, Krupovic M, Yutin N. Evolution of double-stranded DNA viruses of eukaryotes: from bacteriophages to transposons to giant viruses. Ann N Y Acad Sci. 2015 Apr;1341:10-24.
29 Hayes S. F., Burgdorfer W., Barbour A. G. (1983) Bacteriophage in the Ixodes dammini spirochete, etiological agent of Lyme disease. J. Bacteriol. 154:1436–1439.
30 Eggers CH1, Kimmel BJ, Bono JL, Elias AF, Rosa P, Samuels DS. Transduction by phiBB-1, a bacteriophage of Borrelia burgdorferi. J Bacteriol. 2001 Aug;183(16):4771-8.
31Casjens S, Palmer N, van Vugt R, Huang WM, Stevenson B, Rosa P, Lathigra R, Sutton G, Peterson J, Dodson RJ, Haft D, Hickey E, Gwinn M, White O, Fraser CM. A bacterial genome in flux: the twelve linear and nine circular extrachromosomal DNAs in an infectious isolate of the Lyme disease spirochete Borrelia burgdorferi. Mol Microbiol. 2000 Feb;35(3):490–516.
32 Brady JM, Gray WA, Caldwell MA. The electron microscopy of bacteriophage-like particles in dental plaque. J Dent Res. 1977 Aug;56(8):991–3.
33 Miklossy J. Historic evidence to support a causal relationship between spirochetal infections and Alzheimer’s disease. Front Aging Neurosci. 2015 Apr 16;7:46. doi: 10.3389/fnagi.2015.00046. eCollection 2015.
34Bali Z, Bali RK, Nagrath S. Prion diseases: risks, characteristics, and infection control considerations in dentistry. J Investig Clin Dent. 2011 Nov;2(4):236-40.
35 Sushma B, Gugwad S, Pavaskar R, Malik SA. Prions in dentistry: A need to be concerned and known. J Oral Maxillofac Pathol. 2016 Jan-Apr;20(1):111-4.
36 Azarpazhooh A and Fillery, ED Prion Disease: The Implications for Dentistry. JOE—Volume 34, Number 10, October 2008
37 Jayanthi P, Thomas P, Bindhu P, Krishnapillai R. Prion diseases in humans: Oral and dental implications. N Am J Med Sci. 2013;5:399–403.
38 Malmstrom M, Husby G. Occurrence of amyloid in the teeth-supporting tissues of
patients with rheumatoid diseases. An immunohistochemical study. Scand J Rheumatol
1975;4:186 –92.
39 WHO consultation. WHO Infection Control Guidelines for Transmissible Spongiform Encephalopathies. Geneva, Switzerland: World Health Organization Communicable Disease Surveillance and Control; 1999. Report No.: WHO/CDS/CSR/APH/2000.
40 Brown P, Liberski PP, Wolff A, Gajdusek DC. Resistance of scrapie infectivity to steam autoclaving after formaldehyde fixation and limited survival after ashing at 360 degrees C: practical and theoretical implications. J Infect Dis 1990;161:467–72.
41Brown SA, Merritt K. Use of containment pans and lids for autoclaving caustic solutions. Am J Infect Control 2003;31:257.